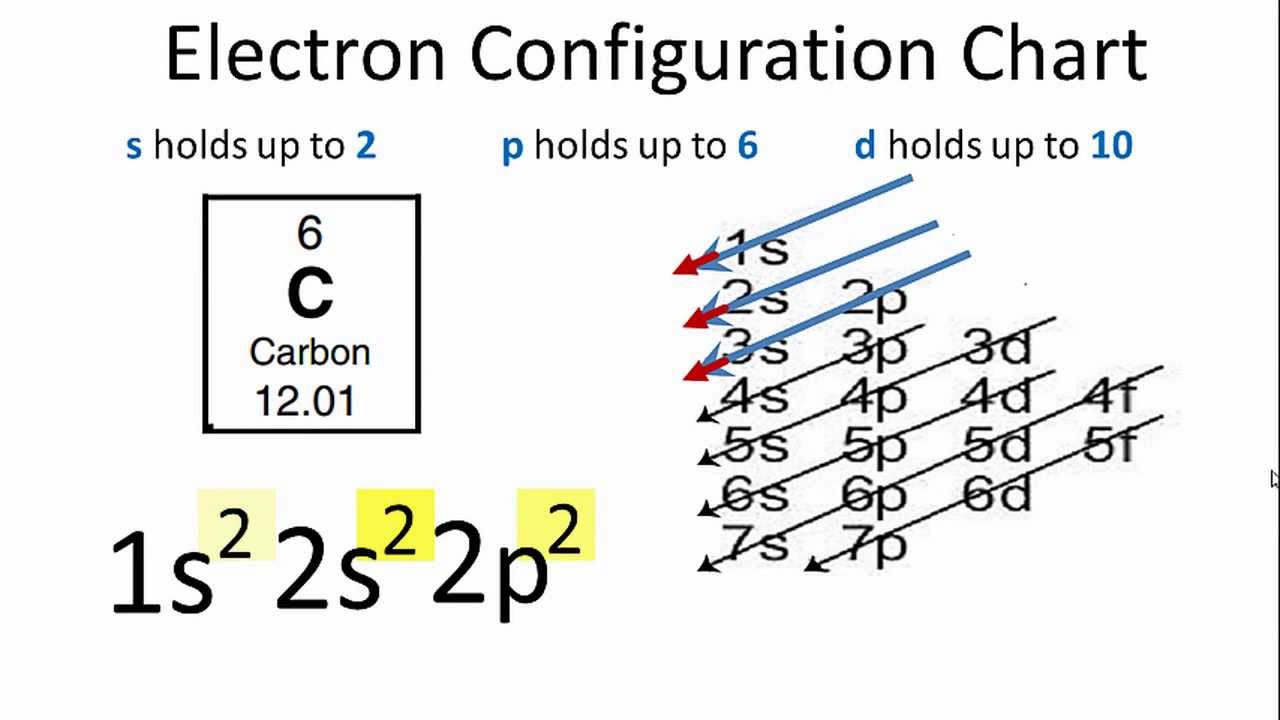
  You will also learn why there are occasional exceptions to this trend and how these exceptions influence elemental properties. Let us take the example of the calcium atom in order to illustrate this concept step by step. The Aufbau diagram combined with the number of electrons that occupy each subshell can determine the electron configuration of many elements 1, 2, 7. Examples of covalent bonding shown below include hydrogen, fluorine, carbon dioxide. Considering the electron configuration for carbon atoms: 1s 2 2s 2 2p 2, the two 2s electrons will fill the similar. In general, an electron will occupy an atomic orbital with the lowest value of n, l,ml n, l. Read more about Aufbau principle and Paulis exclusion principle at. 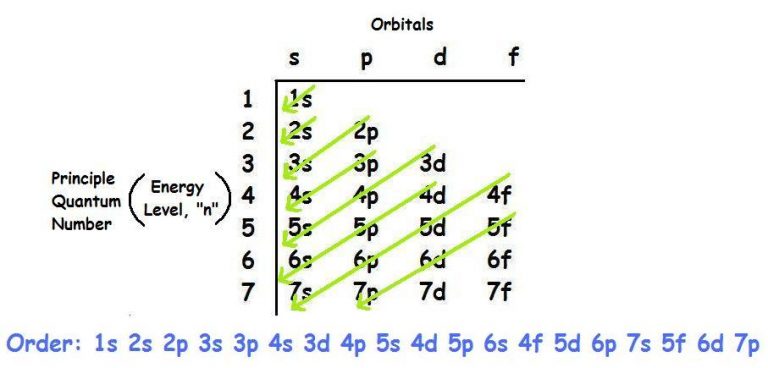 Electrons are filled in atomic orbitals as per the order determined by the Aufbau principle, Pauli. The Aufbau Principle (also called the building-up principle or the Aufbau rule) states that, in the ground state of an atom or ion, electrons fill atomic orbitals of the lowest available energy level before occupying higher-energy levels. In the next section you will learn why the orbital energy levels correlate with shell number and why subshells within a shell usually follow the trend that s of the exceptions are within the d- and f- blocks, and the violations are caused by an unexpected order of the orbital energy levels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
